|
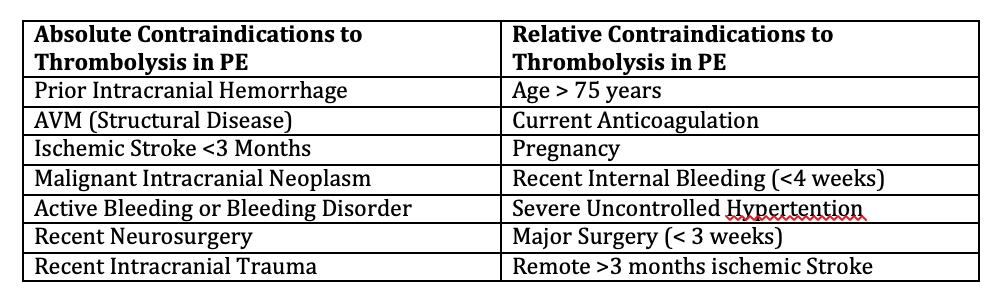
By Emerson Posadas, MD, MBA PGY-3 The use of thrombolysis in the management of pulmonary embolism is controversial. While many physicians will give thrombolytics in patients with massive pulmonary embolism, I have found that many physicians are much more hesitant to utilize thrombolysis for sub-massive pulmonary embolism. While there has been significant evidence behind the use of thrombolysis in CVA and cardiac ischemia, the evidence behind its use in pulmonary embolism is much less clear. Yet, pulmonary embolism represents a significant disease process that is associated with high morbidity and mortality. The AHA and American Cardiology Association in the past year has advocated for the use of thrombolytics in both massive and sub-massive pulmonary embolism in select patients.1However, ACEP has yet to release a clinical policy regarding the use of thrombolysis in sub-massive pulmonary embolism. In this review, I will discuss the evidence behind the use of thrombolysis in pulmonary embolism, specifically in sub-massive pulmonary embolism. In this review, we will assume that the patient already has a diagnosis of pulmonary embolism or likely pulmonary embolism. This review is not to go over the PERC, Wells, or Geneva scores in determining the likelihood of pulmonary embolism. This review guides the clinician on what to do now that the diagnosis of pulmonary embolism has been made. First, it is best to define the different types of pulmonary embolism based on severity. Standard definitions for sub-massive pulmonary embolism have not been universally accepted. However, on review of the literature many studies share similar clinical features that delineate between the different severities. 2Massive pulmonary embolism is generally characterized as associated with cardiac arrest, sustained hypotension (BP <90 or requiring inotropic support), or persistent and profound bradycardia suggestive of shock. Submassive pulmonary embolism is associated with a SBP >90 but with signs of either right ventricular dysfunction or myocardial necrosis. Signs of right ventricular dysfunction include RV dilation on imaging, elevation of BNP (>500), new complete or incomplete RBBB, anteroseptal ST elevation/depression or TWI on EKG. Elevated troponin is a sign of myocardial necrosis. Thrombolysis in general is not indicated in non-massive or sub-segmental PE, in which patients have no evidence of hemodynamic compromise or RV strain.3In some studies, the use of the Pulmonary Embolism Severity Index can also be used to help guide management.4 The MOPETT trial in 2013 studied the use of half dose TPA (0.5 mg/kg max 50 mg) in sub-massive pulmonary embolism. Their main outcome measure was the reduction of pulmonary artery pressure. They found that there was a significant reduction in pulmonary hypertension as well as recurrent pulmonary embolism, however no significant mortality benefit. However, there was no analysis on rates of RV dysfunction or ischemia.5In a separate trial in 2014, the PEITHO trial studied the effect of single dose thrombolysis to reduce mortality or hemodynamic collapse in sub-massive pulmonary embolism at 7 days. They found that fibrinolytic therapy decreased incidence of hemodynamic decompensation, but was associated with up to a 5-fold major bleeding risk. 6 While no single study has had the power to conclusively recommend thrombolytics for pulmonary embolism, there have been several meta-analyses done. A meta-analysis in 2014 found that among patients with pulmonary embolism, thrombolytic therapy was associated with lower rates of all cause mortality but also increased rate of increased bleeding and ICH. Those with RV dysfunction, including those that were hemodynamically stable, seemed to benefit the most. It is important to note that major bleeding was not significantly increased in patients 65 years and younger.7 Absolute contraindications are similar to those seen in the use of thrombolytics for CVA, and are associated with increased bleeding risk. This includes prior intracranial hemorrhage, AVM, suspected aortic dissection, active bleeding, recent neurosurgery, recent trauma with brain injury. Once the provider has decided to give thrombolytics, the question becomes how much? Full dose thrombolytics has been studied in multiple trials including the PEITHO trial (tenecteplase), TOPCOAT trial (tenecteplase), MAPPET-3 Trial (alteplase) which showed consistent decrease in hemodynamic compensation but mixed mortality benefit. Further, there was a increase risk of major bleeding in patients >65 years of age in all trials. 7,8While the MOPETT used ½ dose tPA with improvement of pulmonary hypertension at 28 months with no increase in major bleeding. However, mortality was not assessed in this trial. In one 2010 RCT comparing half-dose vs full dose tPA in patients with pulmonary embolism and hemodynamic instability, researchers found no difference in primary outcomes however there was increased bleeding risk in the full dose tPA group. 9
While more studies need to be conducted, the clinical bottom line seems to be trending towards the use of low dose/half dose thrombolysis in selected patient populations. The use of low dose systemic thrombolysis in patients with sub-massive pulmonary embolism and impending hemodynamic instability seem to benefit the most. There also may be decreased bleeding risk with low dose systemic thrombolysis versus systemic thrombolysis. While all-cause mortality does not seem to change, several other outcome measures do improve. There is no clear-cut answer, and the use of thrombolysis in patients should be made on a case by case basis and also taking into account functional status. It is clear that a component of shared decision-making should be done when considering thrombolysis.
1 Comment
|
ABOUT USVegasFOAM is dedicated to sharing cutting edge learning with anyone, anywhere, anytime. We hope to inspire discussion, challenge dogma, and keep readers up to date on the latest in emergency medicine. This site is managed by the residents of Las Vegas’ Emergency Medicine Residency program and we are committed to promoting the FOAMed movement. Archives
June 2022
Categories |
CONTACT US901 Rancho Lane, Ste 135 Las Vegas, NV 89106 P: (702) 383-7885 F: (702) 366-8545 |
ABOUT US |
WHO WE ARE |
WHAT WE DO |
STUDENTS |
RESEARCH |
FOAM BLOG |
© COPYRIGHT 2015. ALL RIGHTS RESERVED.
LasVegasEMR.com is neither owned nor operated by the Kirk Kerkorian School or Medicine at UNLV . It is financed and managed independently by a group of emergency physicians. This website is not supported financially, technically, or otherwise by UNLVSOM nor by any other governmental entity. The affiliation with Kirk Kekorian School of Medicine at UNLV logo does not imply endorsement or approval of the content contained on these pages.
Icons made by Pixel perfect from www.flaticon.com
Icons made by Pixel perfect from www.flaticon.com

 RSS Feed
RSS Feed
